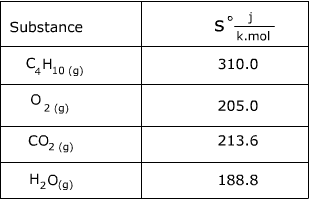
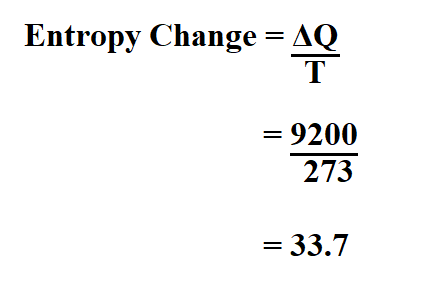How to Calculate the Entropy Change for a Chemical or Physical Process Based on Absolute Entropies | Chemistry | Study.com

Calculate the entropy change in surroundings when `1.00` mol of `H_(2)O(l)` is formed under stan... - YouTube

Calculate the entropy change in surroundings when 1.00 mol of H2 O (l) is formed under standard conditions at 298 K. Given ΔrH^0 = - 286 kJ mol^-1 .

Calculate the entropy change of n-hexae when 1mol of it evaporates at 341.7K (Delta(vap)H^(Theta) = 290.0 kJ mol^(-1))

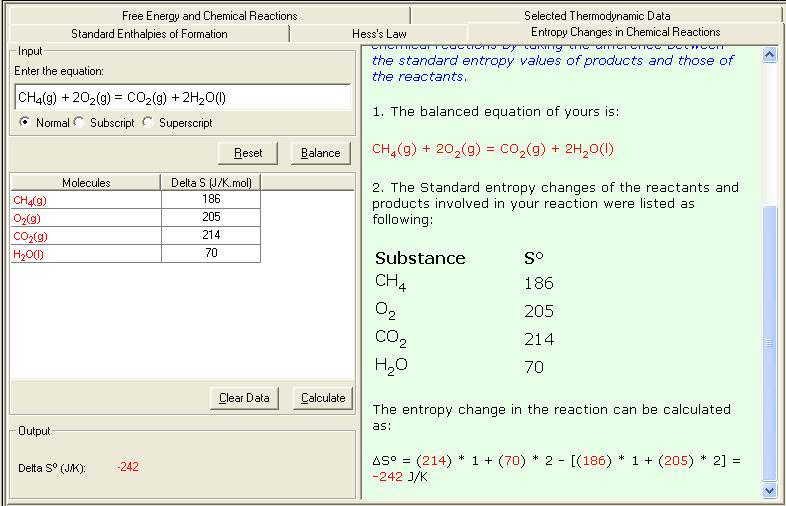
SOLVED: 4 Calculate the standard entropy change for the formation of one mole of nitrogen dioxide from its elements. 'zNz (g) + 0z (g) NOzig) Compound So (J/molek) Nz (g) 191.5 02 (

Calculate the entropy change in surrounding when 1.00 mol of H2O(l) is formed under standard condition fH^ = - 286 KJ mol^-1 .