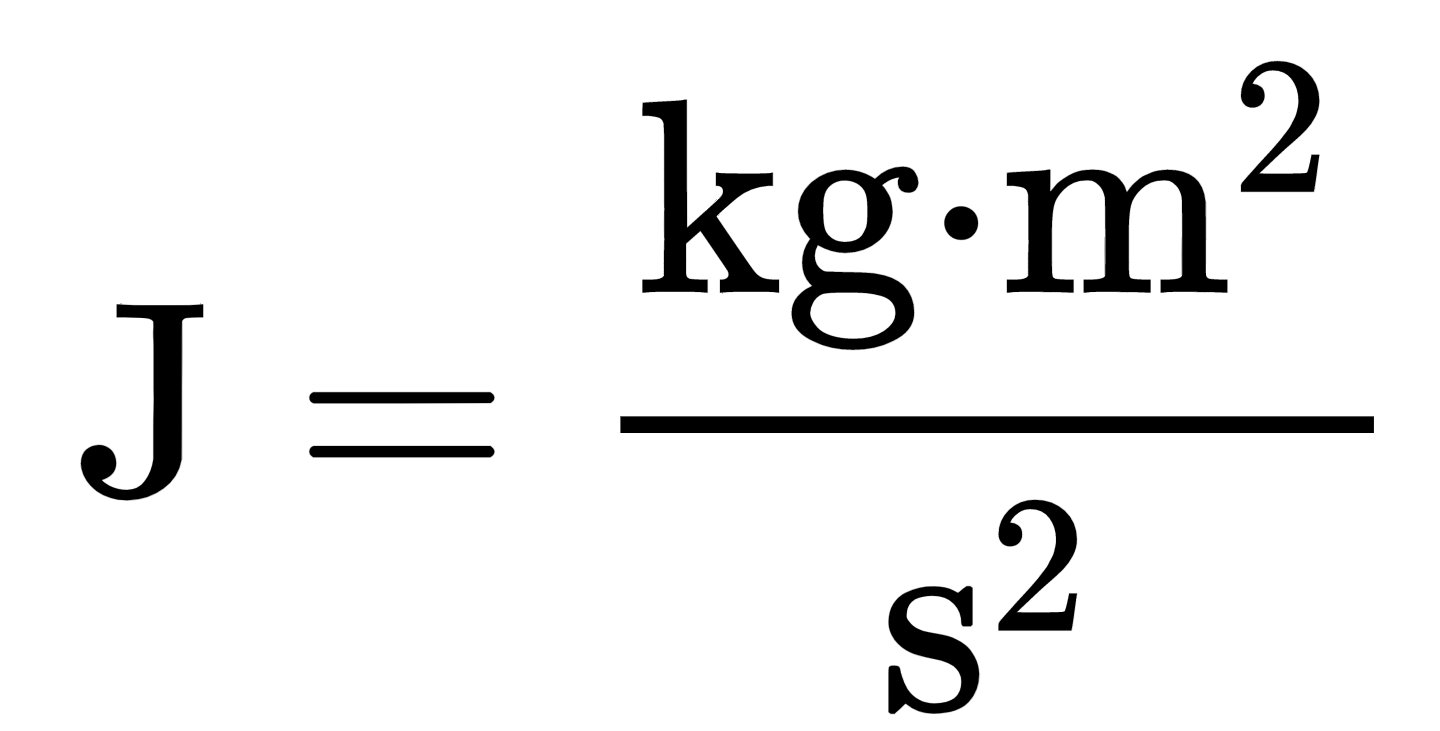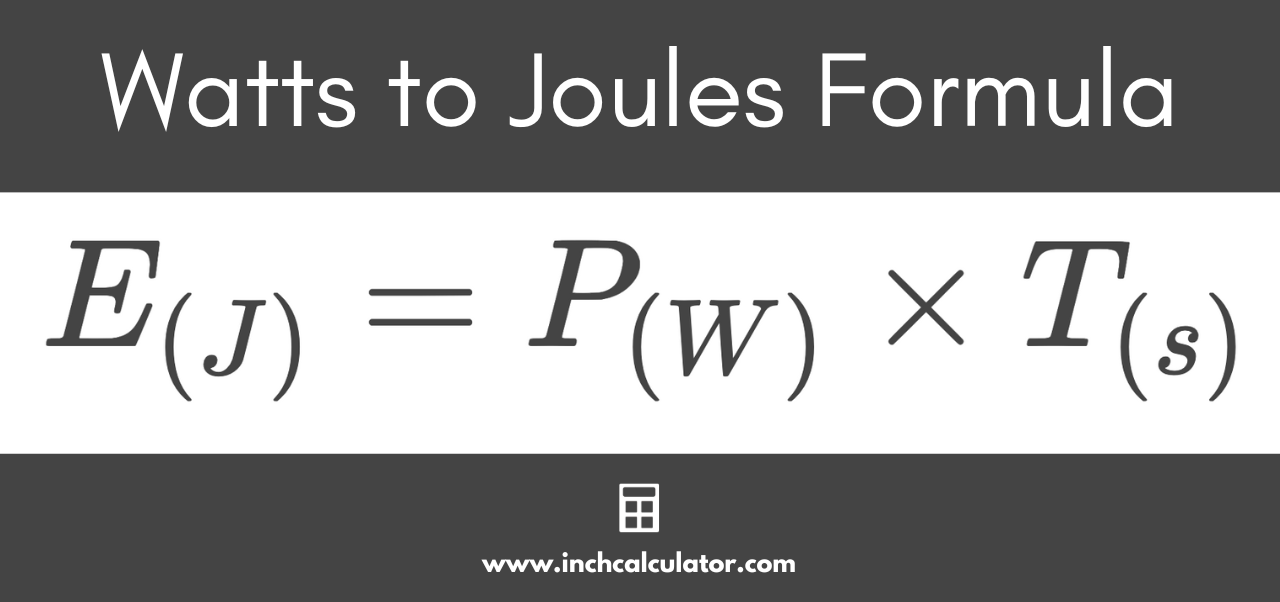1 Given the constants below, calculate how much energy in Joules would be required to change 100.0 g liquid water at 100oC into steam, also at 100oC. SHOW ALL WORK | Wyzant

OpenStax College Physics Solution, Chapter 29, Problem 76 (Problems & Exercises) | OpenStax College Physics Answers