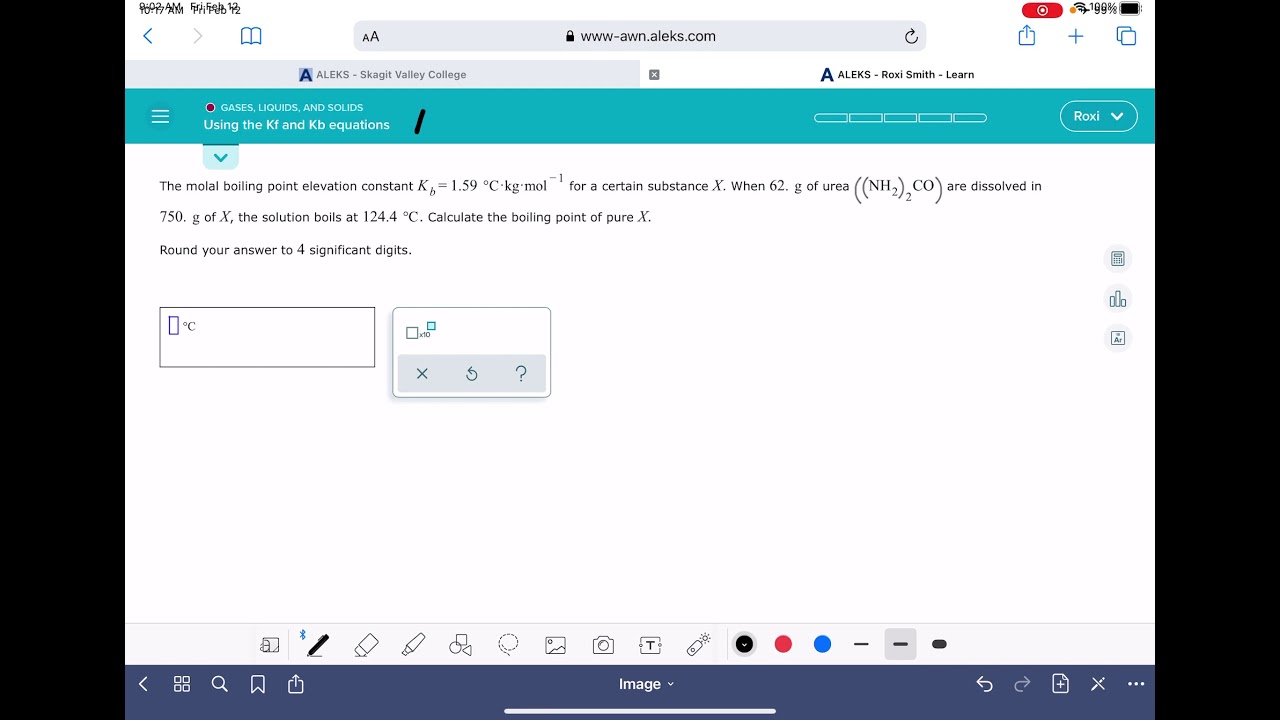
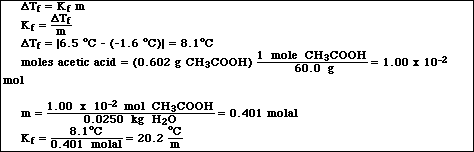Calculate the amount of KCl which must be added to 1 kg of water so that the freezing point is depressed by 2K. (kf for water = 1.86 K kg mol^-1)

How are Kf values relevant in calculations of the melting temperature of a solution? | Homework.Study.com

SOLVED:From the value of Kf listed in Table 17.1, calculate the concentration of Ni^2+ in 1.0 L of a solution that contains a total of 1 ×10^-3 mol of nickel(II) ion and

Calculate Complex Ion Equilibria Using the Small x Approximation for Large Kf | Chemistry | Study.com

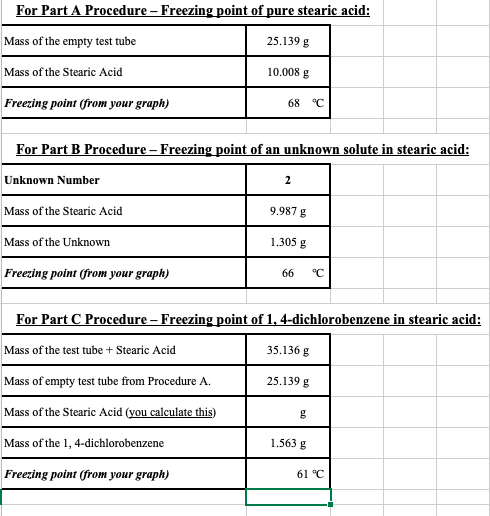
After you preform your experiment, you determine that the Kf value for naphthalene is 6.9 . You are using 10g of naphthalene and added 1.0 g of your unknown. The the freezing

After you preform your experiment, you determine that the Kf value for naphthalene is 6.9 . You are using 10g of naphthalene and added 1.0 g of your unknown. The the freezing

Calculate the freezing point of a solution when 3 g of CaCl2(M = 111 g mol^-1) was dissolved in 100 g of water.assuming CaCl2 Undergoes complete ionisation (Kf for water = 1.86 K kg mol^-1)